Coronavirus trial: People across England start donating plasma
People who have recovered from coronavirus have started donating plasma to help others.
More than 140 people have donated plasma as part of a trial to potentially help people with coronavirus (COVID-19).
NHS Blood and Transplant (NHSBT) is leading a major new programme, on behalf of the Government, to collect plasma from people who have recovered from COVID-19 to support a national clinical trial.
The trial will investigate whether plasma transfusions could improve a COVID-19 patient’s speed of recovery and chances of survival.
In parallel to the trial, we are building up capacity to collect plasma so that we can deliver at a large scale, if transfusions are shown to help patients.
More than 300 people are booked in to donate in the near future.
Dr Abhi Lal, 43 from Solihull, has recovered from COVID-19 and was one of the first people to donate plasma at Birmingham New Street donor centre.
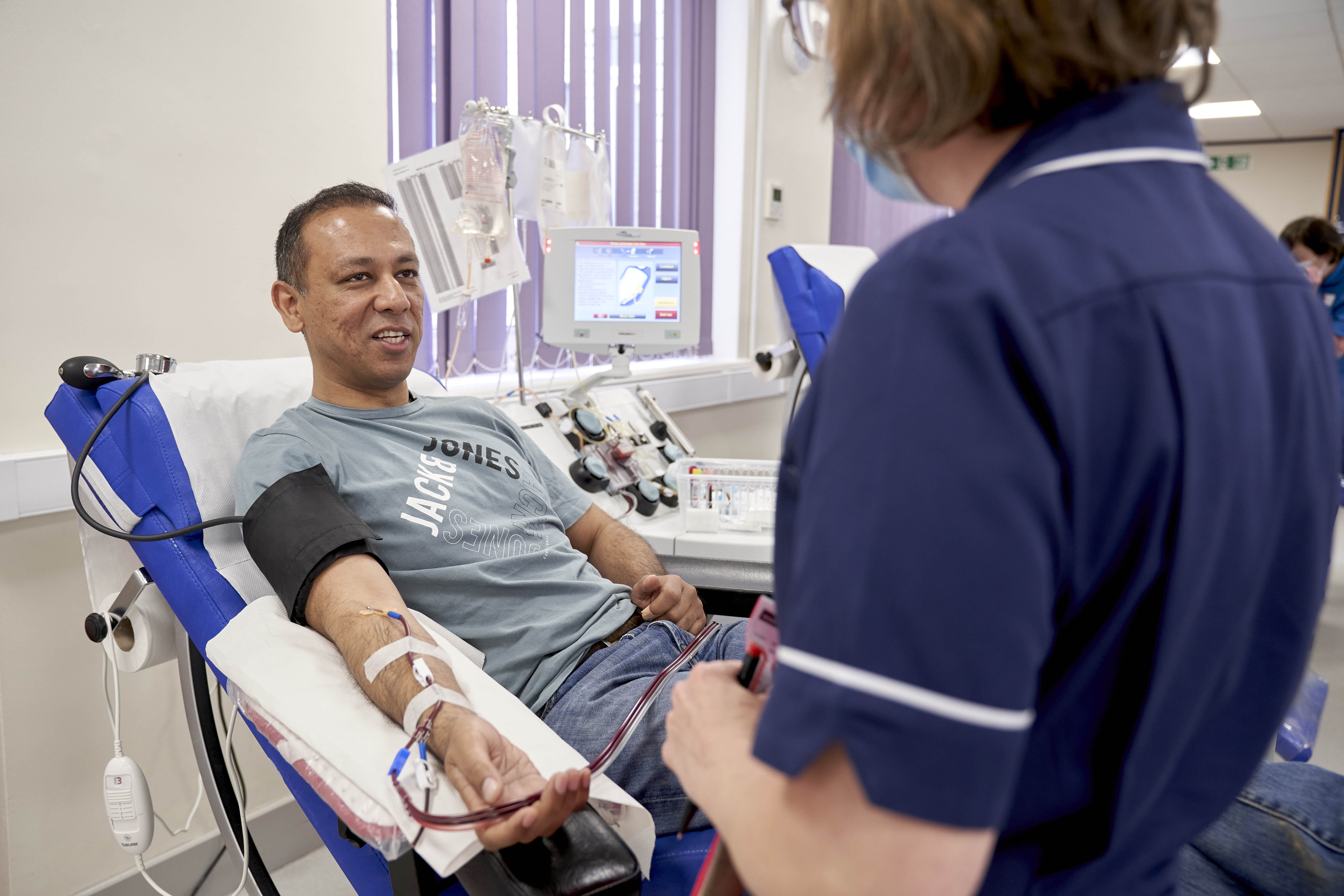 He said: “Donating my plasma was a fantastic experience. The staff have been very friendly, and I have been very well looked after.
He said: “Donating my plasma was a fantastic experience. The staff have been very friendly, and I have been very well looked after.
“I had COVID-19 six weeks ago. I had body aches, fever and fatigue, and a positive test result. I wanted to do something to potentially help others and to help develop a treatment.
An NHSBT spokesperson said: “This trial is world-leading research and people like Dr Abhi Lal are leading the way.
“Plasma donation is safe and easy, and you could help save the lives of others with coronavirus.”
What is convalescent plasma?
Convalescent plasma is plasma from people who have recovered from an infection.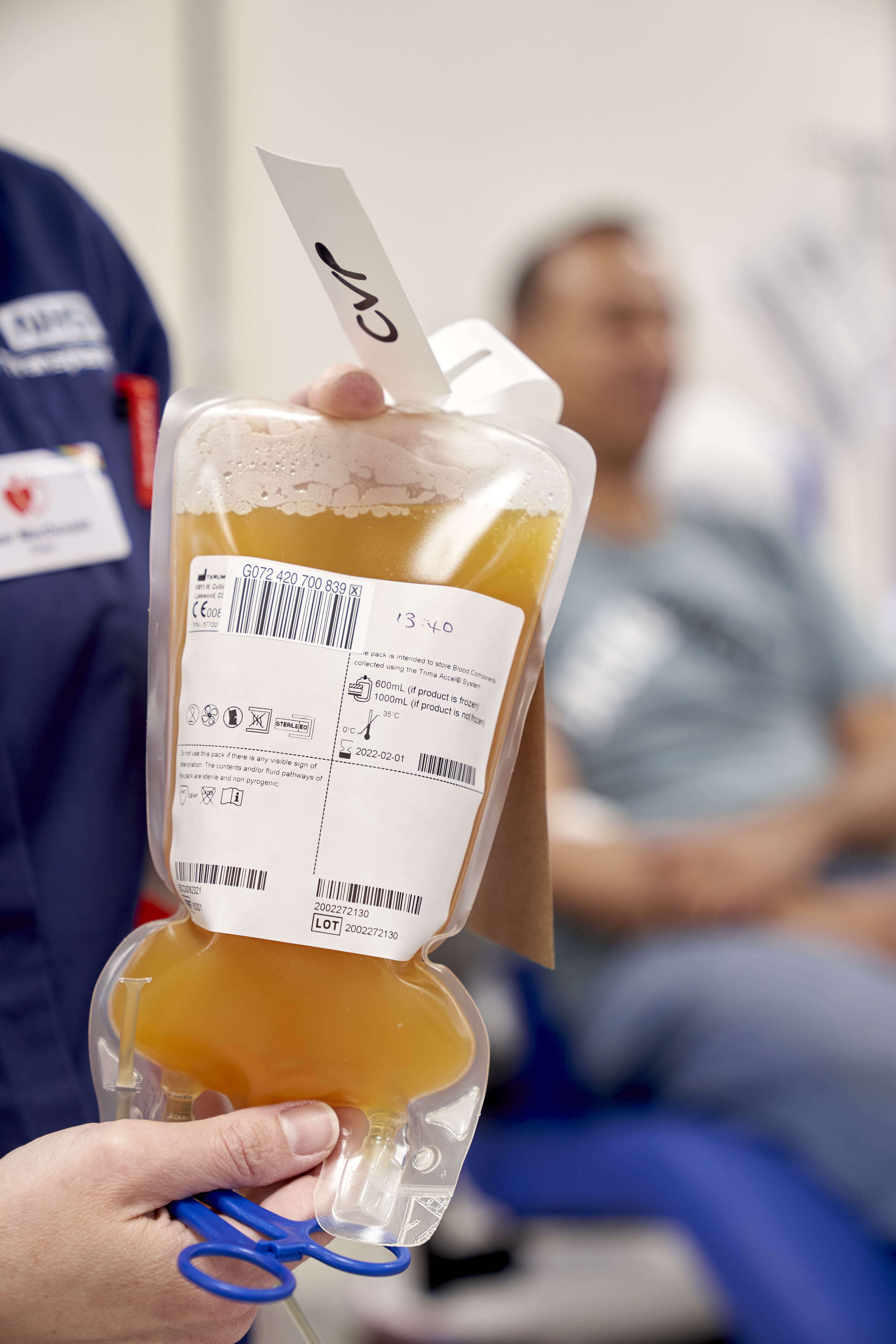
Recovered patients’ plasma may contain antibodies that their immune systems have produced in fighting the virus.
That plasma can be transfused to patients whose immune systems are struggling to develop their own antibodies.
About the trials
The trials will investigate whether transfusions may improve a patient’s speed of recovery and chances of survival. Plasma can also be collected and frozen ahead of any second wave of COVID-19.
Although there is some evidence of patient benefit from the use of convalescent plasma, the safety and effectiveness of convalescent plasma transfusions needs to be confirmed by a robust clinical trial.
Donating plasma
We are mainly contacting potential convalescent plasma donors directly to take part in the trials.
It is important that potential donors have recovered, and their body has had time to develop a good antibody response. For these reasons, we are currently collecting plasma no sooner than 28 days after recovery. This also means that until recently few people were eligible to donate.
The NHSBT spokesperson said: “We recognise some donors will have had a difficult experience and we will make everyone feel cared for and welcome. If you get the call, please donate.”

