Clinical Biotechnology Centre
The Clinical Biotechnology Centre at NHS Blood and Transplant, Filton
A video outlining the Clinical Biotechnology Centre operations at the CBC Filton facility.
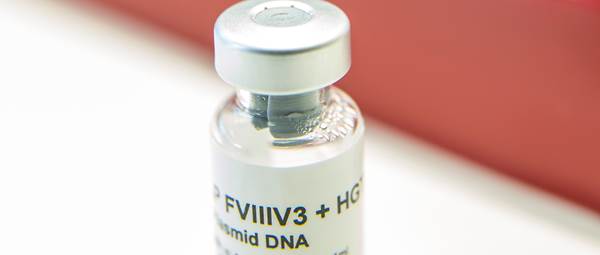
We have been manufacturing GMP-grade DNA plasmids for over 20 years and we now offer viral vectors (lentiviral and adeno-associated viral).
We are proud to have supported numerous clinical trials, treating patients around the world with a broad range of infectious, malignant or inherited disorders.
With a wealth of expertise and experience, we manufacture plasmids for a variety of research, pre-clinical and clinical campaigns, for healthcare, academic and commercial customers.
We have the experience to de-risk your project plans, and the know-how to overcome the challenges of transferring manufacturing processes to GMP compliance for the first time.

 Reasons to choose the Clinical Biotechnology Centre
Reasons to choose the Clinical Biotechnology Centre
- Our track record in small scale production of GMP-grade plasmids and proteins is second to none in the UK
- Our highly skilled staff draw on almost 20 years of experience
- We can manufacture multiple batches of plasmids concurrently on a campaign basis
- We draw on extensive QA and QP expertise across NHS Blood and Transplant to support the production and testing of Investigational Medicinal Products (IMPs) and the preparation of IMP Dossiers (IMPDs)
- We have many years’ experience importing and distributing IMPs manufactured outside of the EU
- We provide a bespoke service, tailored to the different needs of healthcare, academic, start-up and established gene therapy units
- We are an established partner of choice for the world-leading gene therapy groups across the UK and Europe
Milestones